Hybrid hydrogel: from drug release to coral settlement
Abstract

Injectable hydrogels, capable of rapid in situ gelation under physiological conditions, are highly attractive for minimally invasive surgery and locoregional drug delivery [1]. However, the wide majority of polymeric systems rely on UV photo-crosslinking strategies, or slow covalent bond formation limiting in vivo applicability. We have demonstrated the use of hyaluronic acid derivatives as possible matrices because of their fast gelation. Their use as fistula fillers have been demonstrated on model animals (pigs) and their biocompatibility allowed the full healing after 2 weeks [2]. As a biocompatible alternative, we herein report a novel injectable hydrogels composed of gelatin methacrylate (GelMA) and poly(ethyleneglycol) dimethacrylate (PEGDA), crosslinked through a Fenton-like radical polymerization mediated by Cytochrome C (CyC), instead of the iron salt, and in the presence of H2O2 and the biocompatible L-ascorbic acid as reducing agent. The resulting hydrogels, undergoing a sol–gel transition within 1.0 and 2.0 minutes, are biocompatible and their properties are highly tunable. Indeed, rheological analysis showed that mechanical properties and the linear viscoelastic region (LVR) can be easily modulated by varying the concentrations of the starting methacryl-functionalized gelatin and crosslinker [3]. Structural characterization and biodegradation studies revealed that enzymatic degradation can be modulated. The hydrogels is readily injectable and showed no detectable cytotoxicity in conditioned media assays. Sustained release of rhodamine 101, as a drug mimicking system, reached ~70% over 7 days. The hydrogels where also functionalized with “breakable” nanoparticles filed with sorafenib, a well known drug for the treatment of hepatocellular carcinoma. The combination of different release profiles allows the treatment with multiple drugs and the time control of different therapeutics.
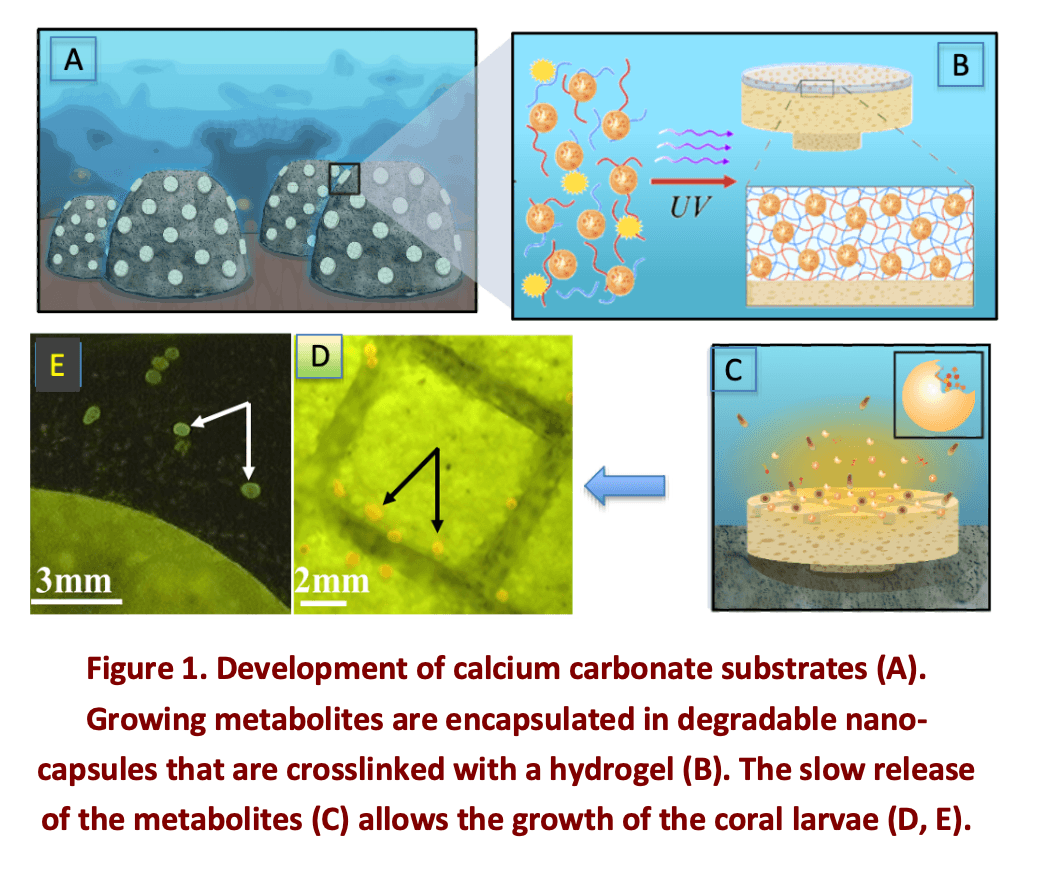
Finally the combination of degradable silica nanocapsules and hydrogels has recently proved to be an exceptional interesting way to to enhance coral settlement through the development of biomimetic microhabitats that replicate the chemical landscape of healthy reefs. The responsive capsules able to entrap large biomolecules are described, and embedded in a hydrogel to slow down the release of the actives.
The concept of the work and the results are summarized in Figure 1. We engineered a soft biomaterial, SNAP-X, composed of silica nanoparticles, biopolymers and algal exometabolites, to enrich reef microhabitats with bioactive molecules from crustose coralline algae. Coral settlement was enhanced over 20-fold using SNAP-X coated substrates compared to uncoated controls. SNAP-X is designed to gradually release chemical signals slowly (> 1 month) under natural seawater conditions, and it can be rapidly applied to natural reef substrates via photopolymerization, further facilitating the light- assisted 3D printing of microengineered habitats[4].
References
1- E. Piantanida, G. Alonci, A. Bertucci, L. De Cola Acc. Chem. Res. 2019, 52, 2101.
2- E. Piantanida, I. Boskoski, G. Quero, C. Gallo, Y. Zhang, C. Fiorillo, V. Arena, G. Costamagna, S. Perretta, L. De Cola Materials Today Bio 2021, 10, 100109.
3- L. Menduti, L. De Cola et al. Submitted.
4- S. Kundu, S.Potenti, Z.A. Quinlan, H. Willard, J. Chen, T.Noritake, N. Levy, Z. Karimi, H. Jorissen, J.R. Hancock, C. Drury, L. Wegley Kelly, L. De Cola, S. Chen, R3D consortium, D. Wangpraseurt Trends Biotechnol. 2025, 43, 2232.